Bio Farma Chief Outlines Indonesia's Health Product Opportunities Following BPOM's WHO Listed Authority Status
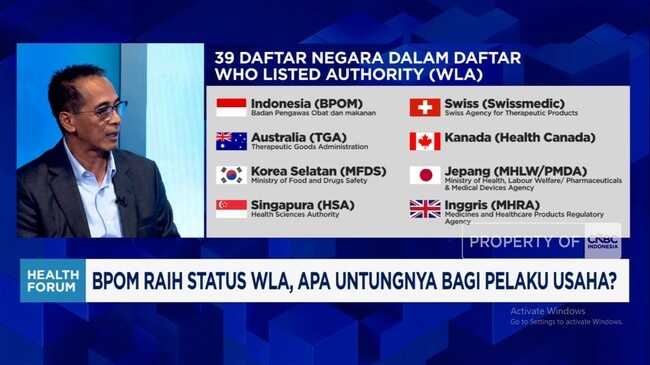
Jakarta, CNBC Indonesia - PT Bio Farma (Persero) has stated that Indonesia has an opportunity to accelerate the development of new health products whilst strengthening exports of such products. This is supported by the success of the Indonesian Food and Drug Authority (BPOM) in achieving WHO Listed Authority (WLA) status.
Bio Farma Chief Executive Officer Shadiq Akasya expressed appreciation for BPOM’s WLA status achievement. This status indicates that the quality equivalence of national health products is now also recognised at the international level. In fact, some Bio Farma products already adhere to quality standards based on WHO Prequalification (PQ-WHO) from the World Health Organization.
“And this (WLA) represents for us an acceleration for the future, where not only existing products but also products we will develop and export in the future will become easier,” he said during a Health Forum with the theme “BPOM Achieves WLA Status, What Benefits for Business Operators?”, reported Monday (2 March 2026).
According to Shadiq, the WLA status does not only impact Bio Farma as a state-owned pharmaceutical enterprise, but the entire pharmaceutical industry ecosystem in Indonesia. The national pharmaceutical ecosystem is expected to upgrade with the ease of domestically produced products being accepted abroad.
With the WLA status, the export process for health products will be more efficient. In this regard, business operators need only compile certain documents required for the assessment process. In addition, several direct inspections will still be conducted by the WHO.
Furthermore, the existence of WLA is also expected to encourage the national pharmaceutical industry to become more self-reliant by reducing dependence on significantly large imports of raw materials.
“If we aspire to achieve health independence as has been stated, this is very possible with the WLA status,” he concluded.
